- Home
- Research
- Nanoscopy
- Research results
- Nanoscale catalysis
Nanoscale catalysis
10.03.2015
Catalytic conversions generally take place in a very localized manner – atomic steps on metals and reactive centers in proteins are typical examples here. Using a plasmonically activated protonation, it was possible for the first time to carry out a bimolecular reaction on a nanoscale under ambient conditions. This opens up new avenues for reaction control and analytics.
By: VOLKER DECKERT, P. SINGH, TANJA DECKERT-GAUDIG, Z. ZHANG
Heterogeneous catalysis is ubiquitous. The exhaust gases of every motor vehicle are cleaned by the “catalyst” and also in the chemical industry almost every process becomes more energy efficient by the use of catalysts. An interesting development in recent years is the use of metal particles that can be specifically activated by plasmon resonances, i.e. by light. This not only allows reactions to be better controlled in terms of energy, but also makes it possible to guide the reaction in a site-selective manner by specifically directing the light. The latter is known, for example, in a similar form from the exposure of microchips or the like, but without the use of catalysis here so far.
The plasmon assisted catalysis allows, in contrast to the classical exposure, a spatial resolution which is in principle only limited by the dimensions of the active metal particle. In this way, nanometer-scale patterning is possible. At the same time, the particles can also serve as an analytical probe, since the same properties of the nanoparticles are used for so-called tip-enhanced Raman scattering (TERS), which provides structural information with a spatial resolution of less than 5 nm. In this way, both reaction and simultaneous reaction control are possible.
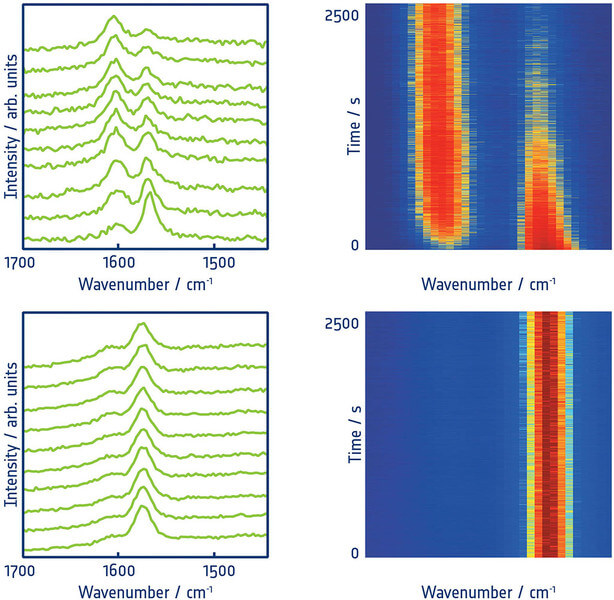
Fig_01 shows the plasmon activated protonation of p-thiopyridine – i.e. a bimolecular reaction. In this model reaction, a single p-thiopyridine layer was deposited either on a silver island layer for macroscopic studies, or on a gold single crystal for the TERS experiments. This allowed controlled conditions to be created and enabled unambiguous evaluation of the results.
The ability to perform structure-relevant analysis simultaneously with the actual reaction clearly shows that the original thiopyridine is completely protonated within a period of several minutes. Interestingly, no acid was added, so the necessary proton must be supplied from the environment. This is proven by further experiments under inert gas – here no further conversion occurs and no protonation products could be detected. The experiments work both macroscopically, i.e. on rough surfaces, and under TERS conditions, when only a single catalytically active nanoparticle is used and irradiated with light.
The experiment strongly demonstrates that plasmonically activated catalysis with nanoscale spatial resolution is possible and, in particular, that complex reactions can be carried out. Thus, applications in nanoscale lithorgraphy as an alternative to expensive electron or ion beam patterning are conceivable.
Funded by: DFG, TAB, Alexander von Humboldt Foundation.
