- Home
- Research
- Biophysical Imaging
Scientific Profile
To better understand living matter such as cells and therefore to better treat diseases, we need to understand how molecules and cells interact with each other. Importantly, we need to understand the physics behind it, i.e. the biophysics. For this, scientist rely on highly sensitive and non-invasive analysis techniques, including far-field fluorescence microscopy. The aim of our Super-Resolution Microscopy Group is providing optimized observation techniques to in more detail understand molecular and cellular interactions especially on membranes. To do so we need to learn about the shortcomings of existing techniques and how to overcome them. The group is doing so through several collaborative cell-biological and biomedical research projects.
More information at: www.biophysical-imaging.com
Research Insights
Unfortunately, this video can only be downloaded if you agree to the use of third-party cookies & scripts.
Research Topics
- Fluorescence microscopy with high spatial and temporal resolution
- Observation of molecule diffusion dynamics (fluorescence correlation spectroscopy, single-molecule tracking)
- Methodological improvements (tissue images, adaptive optics, combination of methods, correlative recording)
- Advanced data analysis tools
Areas of application
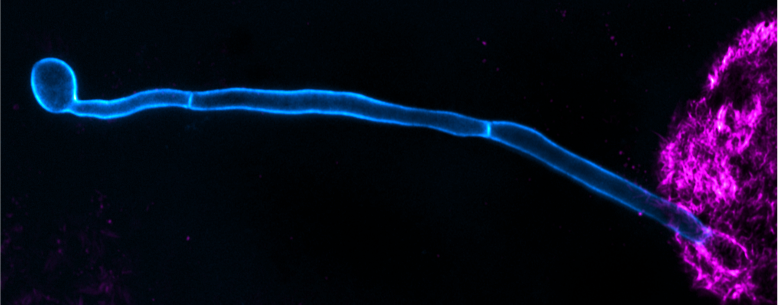
- Investigation of molecular diffusion/interaction dynamics in membranes (lipids, receptors, “lipid rafts”) and biological relevance (e.g. immune cells) using single-molecule fluorescence spectroscopy tools (FCS, tracking) and super-resolution microscopy (STED)
- Microscopy: STED, FCS, Tracking, MINFLUX, iSCAT, …