- Home
- Technology Groups
- Competence Center for Micro- and Nanotechnologies
- Research results
- On-chip Spectroscopic Assessment of Microbial Susceptibility to Antibiotics within 3.5 Hours
On-chip Spectroscopic Assessment of Microbial Susceptibility to Antibiotics within 3.5 Hours
23.04.2018
An integrated microfluidic device is presented which locally enriches bacteria from diluted suspensions (e.g., urine) for subsequent, detailed Raman-spectroscopic characterization. In this way, information about the antibiotic resistance is available within 3.5 hours. This could help the treating physician prescribe tailored, narrow-band antibiotic therapy to the patient.
By: Ute Neugebauer // Ulrich-Christian Schröder // Johanna Kirchhoff // Uwe Glaser // Uwe Hübner // Günter Mayer // Thomas Henkel // Wolfgang Fritzsche // Jürgen Popp
In times of rising antibiotic resistances, new and precise methods which can determine antibiotic susceptibility of infection-causing pathogens within a few hours are urgently needed. While established methods in routine microbiological practice are very accurate and cost-efficient, they are based on the cultivation of bacteria from the patient’s sample. Thus, it requires at least one day, and more often up to two or three days. Cultivation is necessary in order to generate sufficient biological material of the bacterial pathogen for subsequent tests.
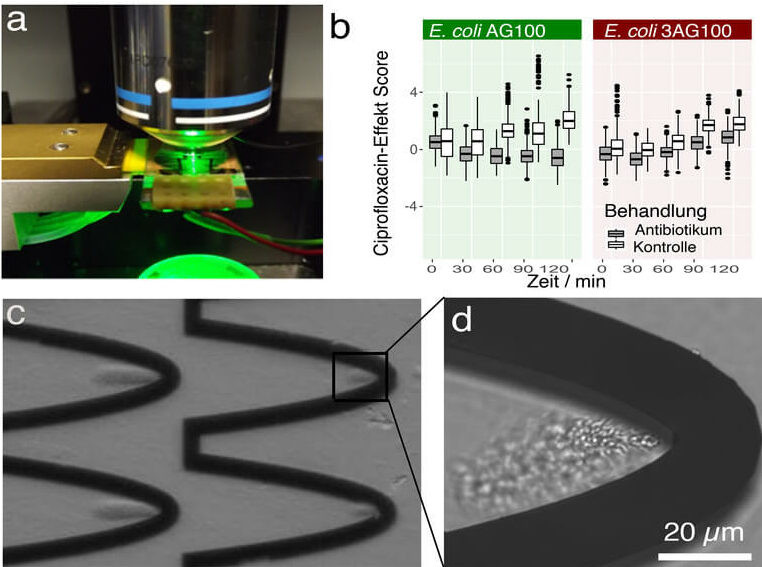
Here, we report on an integrated microfluidic device (Figure 1a) which enables capturing of bacteria directly from diluted suspensions, such as the patient’s urine samples, and allows for subsequent, detailed Raman spectroscopic characterization of the pathogen, including its antibiotic susceptibility, in a label-free and non-destructive manner (Schröder et al. J. Biophotonics 2017). Advantages of this microfluidic device include the minimum sample preparation required prior to loading the sample onto the chip, automated sample processing on the chip, which means minimum hands-on time for the operator and minimal interaction time with infectious material. At the same time, accurate results can be obtained from spectroscopic analysis within very short times, thus speeding up microbial susceptibility testing to identify resistant strains within only a few hours (< 3.5 hours including all preparation steps).
To carry out on-chip analysis, a small amount (< 100 microliters) of the bacterial suspension is loaded into syringes and automatically pumped into the microfluidic chip. In the analysis chamber, sinusoidally-shaped electrodes (Figure 1c) generate an inhomogeneous electric field. A dielectrophoretic force acts on the bacterial particles, which are pumped through the chip. While the liquid continues to flow through the chip, the bacteria are held back in the tip region of the wave where they accumulate (Figure 1c, which is enlarged in Figure 1d). Automated medium exchange is possible, which might be necessary in order to avoid a strong fluorescent background in the Raman analysis caused by varying solutes in the patient’s urine sample.
The bacterial cloud (Figure 1d) is now in the focus of the laser used for Raman characterization. High-quality Raman spectra can be obtained within 1 s / spectrum. In order to gain a representative overview, 300 spectra in six different capturing cavities have been collected. In order to identify resistant bacteria, their Raman spectra in the presence and absence of the respective antibiotic have to be compared. Exemplarily, this was shown for Escherichia coli, the most frequently encountered pathogen in urinary tract infections, and the commonly prescribed fluoroquinolone drug ciprofloxacin. Figure 1b shows the different behavior of sensitive E. coli (E. coli AG100) and resistant E. coli (E. coli 3AG100) with respect to the ciprofloxacin effect score which was calculated from the Raman data. While treated (gray) sensitive bacteria show a negative ciprofloxacin effect score, resistant bacteria have a – like the untreated control group (white) – positive score. A clear differentiation is possible even just after sixty minutes of interaction time.
Often the treating physician and infectiologist is not only interested in whether the pathogen is sensitive or resistant against a particular antibiotic but also wants to know the minimal inhibitory concentration (MIC). This is the lowest concentration of the drug that prevents visible growth of the bacteria. The gold standard method to determine the MIC is an overnight broth microdilution assay. In routine clinical practice, often automated laboratory machines (such as e.g. Vitek 2 from bioMérieux and Phoenix from BD) are used, which also usually take around eight hours to produce results. Raman spectroscopy holds the potential to speed up this analysis, as was recently demonstrated in the example of E. coli and ciprofloxacin (Kirchhoff et al., Anal. Chem.). An algorithm was presented that allows the determination of MIC in less than two hours and was successfully applied for the characterization of several clinical isolates from sepsis patients.
Funded by: BMBF, Thuringia, EU
