- Home
- Research
- Nanobiophotonics
- Research results
- Metal nanoparticles of every shape and color
Metal nanoparticles of every shape and color
19.08.2016
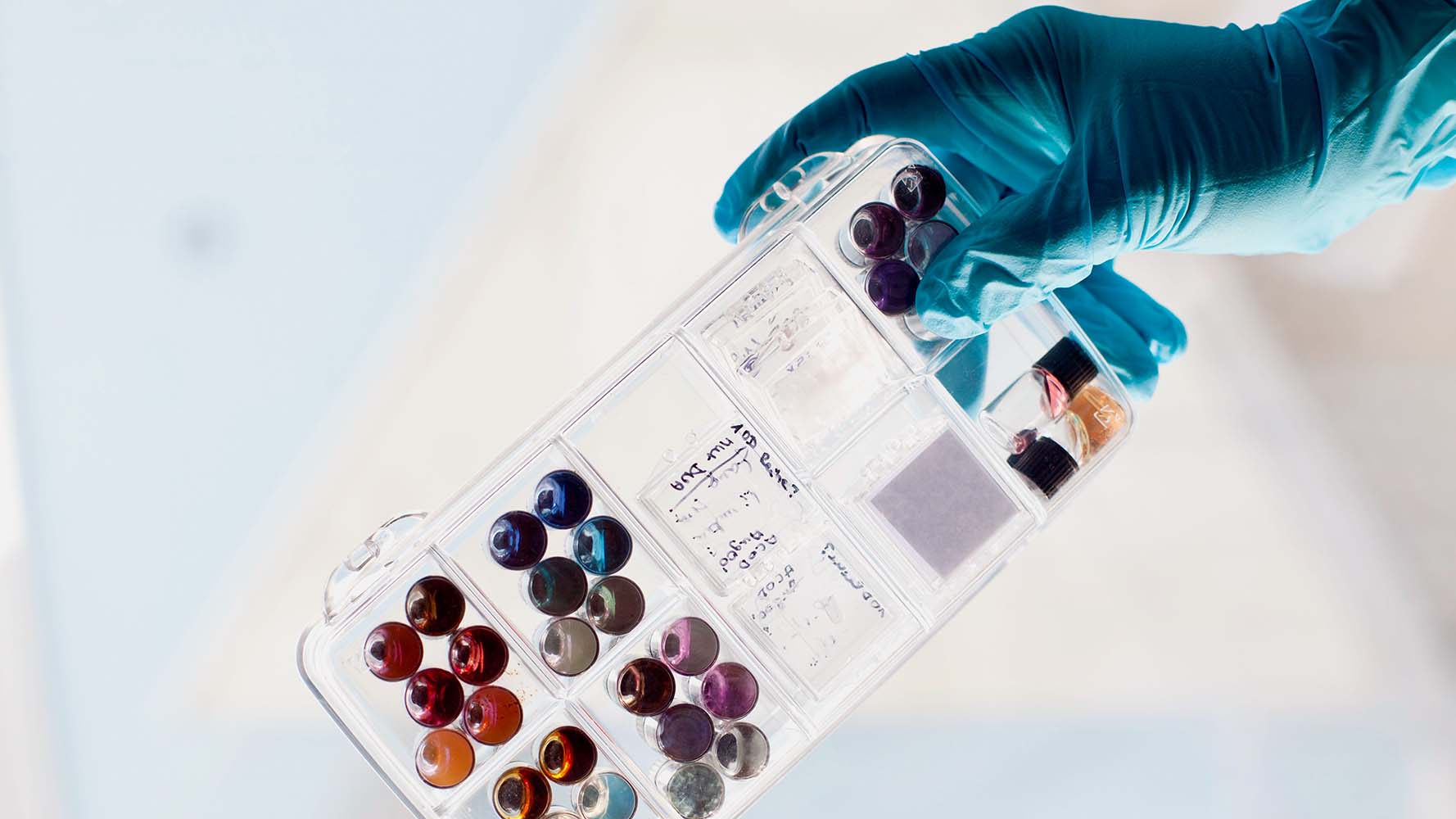
In ancient times, people were already using gold nanoparticles to produce richly colored glass for drinking vessels and jewelry. The intense color is created through the interaction of the metallic nanostructures with incident light, which excites coherent vibrations in the conduction electrons in the metal. Since the resonance frequency of these plasmons is mostly within the visible spectral range, depending on their size, shape, material, and environment, the particles absorb and scatter different wavelengths of light and consequently have different colors. With customized metallic nanoparticles, scientists can now accurately control the position of localized surface plasmons and their interaction with light. As optical markers for biomolecules, signal transducers in sensor technology, or optical antennae, plasmonically active nanoparticles provide an outstanding means for the solution of bioanalytical questions.
Leibniz IPHT consequently uses a nanotechnological bottom-up approach to produce gold, silver, platinum, and palladium nanostructures. In this typically multi-stage process, a reducing agent initially converts metal ions in a solution into elemental metal clusters. These aggregate to produce crystal nuclei approximately three to four nanometers in size, known as seeds. In the subsequent step, the crystal nuclei grow in the presence of a weaker reducing agent and metal salt to form nanoparticles of different shapes and sizes. In addition to generating energetically favorable spherical particles, the forced growth of one or more facets of the crystal nucleus produces nanoscale prisms, cubes, rods, and stars. Their anisotropic shape enhances the plasmonic field at specific points on the surface of the particle. Without this field, the high-sensitivity detection of minute refractive differences, the basis for the most frequent sensory applications of plasmonic nanoparticles, would not be possible.
The plasmonically active nanostructures are usually produced using a batch approach, in other words, in a reaction vessel. However, complex anisotropic structures such as large numbers of high quality nanocubes and nanoprisms can be synthesized more efficiently in microfluidic reactors. These reactors allow for the precise manipulation of crystal nucleation and growth, which have very different reaction kinetics and should normally occur apart from each other both spatially and temporally. The ability to set optimal parameters for each reaction and to accurately control them allows for the production of reproducible particles with a highly uniform shape and size. Particularly in the critical step of crystal nucleation, the short diffusion lengths and accompanying high mixing rates within the small channels of the microfluidic reactor ensure the necessary efficient and homogeneous mixing of the chemical substances.
